The rising demand for plant-based nutrition world-wide requires the deployment of pesticides, in order to protect the crops and plant products from harmful organisms. Safe and high-quality harvests are not possible without targeted plant protection measures. On the other hand, it is a proven fact that large-area deployment of pesticides has caused a significant reduction of the insect population. The decrease of the wild bee population is especially problematic, since wild bees are significant pollinators, and as such an essential prerequisite for agricultural production.
Insect cells as sensors for environmental toxins

Pesticides shall become more „bee-friendly“
The obvious solution to this dilemma between necessary plant protection and insect protection is the development of pesticides without insecticidal effects. The Fraunhofer EMFT scientists support the development of such „bee-friendly“ pesticides by doing research on sensor technologies identifying „bee-harmful“ substances in early phases of pesticide development. The sensors are supposed to detect the insecticidal effects within hours, due to their extremely short reaction times, while enabling parallel analysis of small substance libraries.
Insect cells as sensors

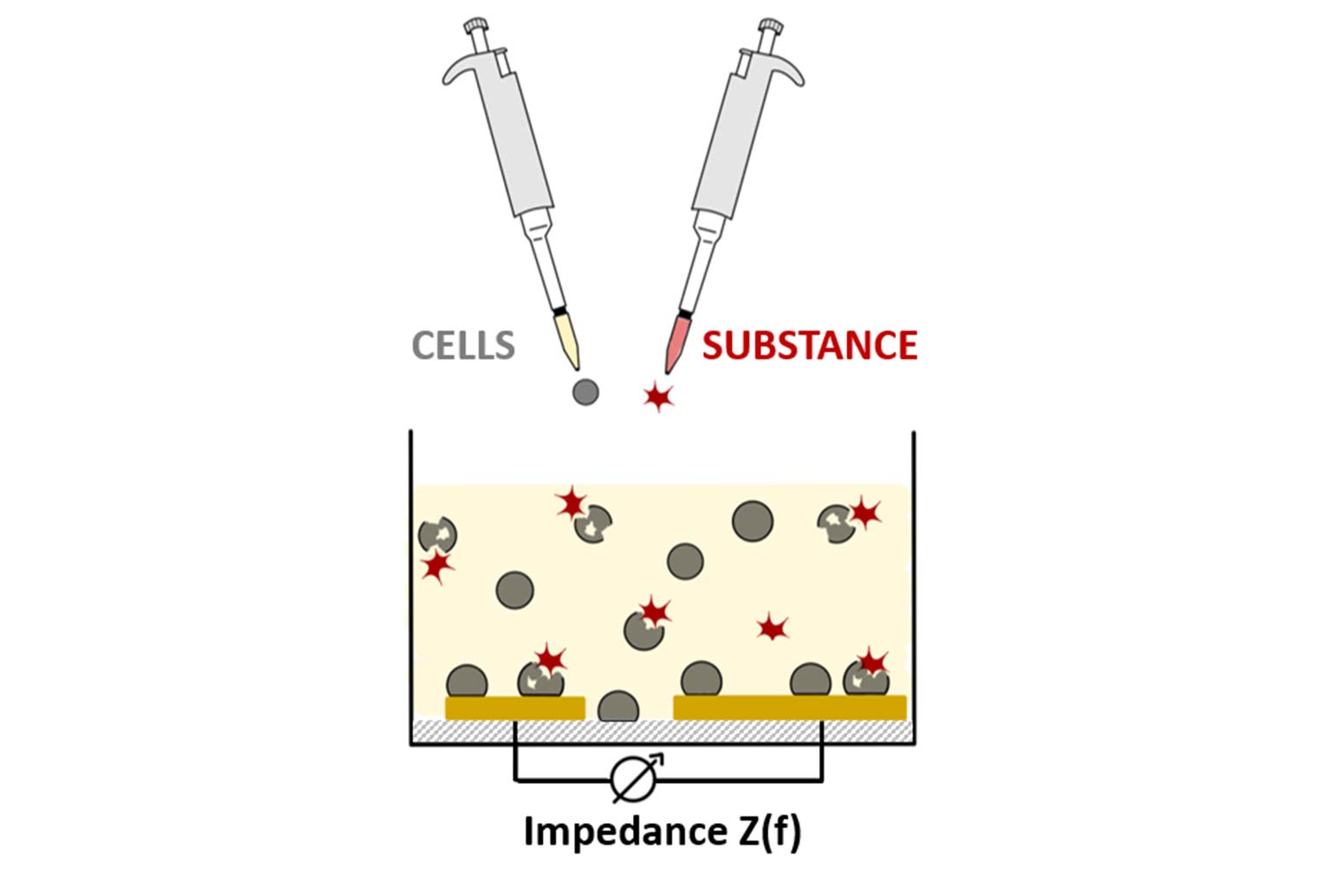
The concept is based on the use of insect cells as sensors. The cells isolated from insects can be multiplied in the lab indefinitely, and are thus available in unlimited amounts. The cells are only few micrometers in diameter but still possess the metabolism characteristic to the insects they were originally isolated from. Exposed to a potentially harmful substance, they show how it affects cell metabolism. In order to measure these effects, the suspended, isolated insect cells are placed in a vessel with microelectrodes prepared on its bottom. These electrodes enable real-time electrochemical impedance measurements monitoring the time course of cell adhesion on the electrode surface. The adhesion process is very sensitive to harmful interferences affecting the cell metabolism, allowing for fast identification of insecticidal effects without lengthy lab times. Unlike typical biosensors, which are used to measure the concentration of a substance, this approach measures the biological impact of the substance on the cells of a certain insect species (dose-response relationship). This approach is basically applicable to cells of various insect species, so that the use of multielectrode arrays will enable the parallel assessment of insecticidal effects of a candidate compound on various insect species in the future.
Current developments
For future in-field applications of the assay technology, decoupling of cell culture work from the actual cytotoxicity assay is mandatory. Thus, suspended, isolated insect cells are frozen directly inside the wells of multielectrode arrays and the cell-loaded electrode arrays are stored at very low temperature. The cells can be thawed anytime for conducting the assay. The cell-based biosensor is assay-ready immediately after thawing the cells within minutes, providing independence of a cell culture laboratory or a laboratory environment in general. This enables a time and cost-efficient preparation of batches of sensor cells weeks or even months prior to the time point when the assay is conducted.
Five different pesticides were tested so far with this optimized sensor technology for their concentration dependent cytotoxicity (dose-response curve), expressed as concentration of half-maximum impact (EC50), by impedance measurements. Validation against a commercial, well-established cell viability assay reveals good agreement with respect to the concentrations of half-maximum impact. Furthermore, data are very much in line with the use of “fresh” cells that were not frozen prior to the assay. Our studies reveal cytotoxic effects of some pesticides sold for home usage for concentrations far below the recommended concentration of use and are found to be more toxic than formulations sold for agricultural industry only.
A benchtop device that performs thawing of cells and sample addition was developed by the Fraunhofer EMFT research team to fully automate the handling of the described cell-loaded electrode arrays. Therefore, the cubic 32.4 cm x 31.4 cm x 31.1 cm small device provides active heating, a holder for two electrode arrays, and a linear robotic system to move the latter in cartesian coordinates. This movement enables the system to fill every well of the electrode array with a pesticide solution of well-defined concentration via a dosing needle. All relevant parameters can be adjusted via a touch screen directly on the housing.
The fluid is applied by free jetting such that the needle is never in contact with the medium in the wells, thus avoiding any cross-contamination. An accurate dosing for a variety of dilutions is achieved by applying an Fraunhofer EMFT-built metal micro pump.
Demonstrator for bee-friendly pesticides
Outlook: Complete efficacy profile instead of mere toxicity measurement
Besides providing basic proof of an insecticidal impact, the sensor technology presented here shall be applicable for multiparameter analysis of various cell functions (phenotypes) later in the project. Through modifications in the assay work flow and by applying different AC frequencies in the measurement, the impedance signals reveal changes in cell proliferation, cell migration or cell motility. This would enable the compilation of a complete efficacy profile of the substance, beyond sole measurement of toxic effects.